  
The SCC susceptibility of E690 steel increases with the positive shift of applied anodic potentials, attributed to the acceleration of anodic dissolution (AD) effect under weak anodic polarization and synergistic effect of AD and hydrogen embrittlement (HE) under strong anodic polarization. 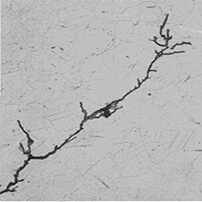
Pitting corrosion initiated around complex oxide inclusions and its transformation to uniform corrosion dominates the corrosion evolution process at OCP, while local acidification induced corrosion pits beneath thick and cracked rust layer dominates the localized corrosion under strong anodic polarization. Composition of corrosion products is not altered by anodic polarization but the rust layer morphology and structure are changed. Positive shift of anodic potential accelerates the two dissolution stages including electrochemical oxidation from Fe to Fe 2+ and Fe 2+ to Fe 3+ of E690 steel. As a final result, the authors propose the key points of a qualitative failure model which consists of a first step of accumulation of Sn and Pb in crack tips, a second step of production of FeSn compound, and a final step of FeSn cracking due to accumulated stresses.In the present work, corrosion evolution and stress corrosion cracking behavior of E690 steel for marine equipment and construction under potentiostatic anodic polarization are investigated via electrochemical analysis, morphology and topography observation and slow strain rate tensile tests. Microscopy revealed intergranular propagation and the existence of an intermetallic compound, the FeSn, at the crack tips. The failure assessment diagram (FAD) of the steel for two different load conditions. 
The subcritical crack propagation rate (d a/d t) II and 3. The value of the threshold stress intensity factor, K th, which is the lowest value to produce cracking 2. Besides, crack characterization tests on steel CT specimens which are immersed in liquid galvanization bath with Zn, 1.1% Sn and 0.1% Bi at 450 ☌ are detailed. Hydrogen influence is discarded after the hydrogen concentration tests performed in this paper. Traditionally two processes have been related with this phenomenon: liquid metal assisted cracking and hydrogen embrittlement. The AD stress corro-sion mechanism includes the slip dissolution mechanism, oxide film cracking mechanism, selective dissolution mechanism, and corrosion-promoted plastic deformation mecha-nism 4,5. Stainless steels resistance to corrosion results from the chromium, which forms a passive film that can protect the material and self-heal in the presence of oxygen. Failures during hot-dip galvanizing are occasionally, but they are important because of the high responsibility of the evolved structures. Stainless steel is an alloy of iron that is resistant to rusting and corrosion.It contains at least 11 chromium and may contain elements such as carbon, other nonmetals and metals to obtain other desired properties. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
